Evolution of proangiosperms during Late Triassic: pre-Cretaceous pollen trends towards mono- and dicotyledonous taxa diversification
*G. Vasanthy, **B. Cornet and ***S.A.J. Pocock
*Institut Français, RB. 33, Pondicherry-605
001, India
**27 Tower Hill Ave., Red bank, New Jersey, USA
*** R.R. 1 West Arrow Creek, Creston, B.C., Canada
Vasanthy, G., Cornet, B.& Pocock, S.A.J. 2004. Evolution of proangiogperms during Late Triassic: pre-Cretaceous pollen trends towards mono- and dicotyledonous taxa diversification. Geophytology 33(1&2): 99-113.
The Late Triassic angiospermid palynomorphs (mostly from the Chinle Formation, Arizona and the Richmond basin, Virginia, USA), Equisetosporites chinleanus Daugh., Cornetipollis reticulata Pocock & Vasanthy and the Crinopolles group (Cornet, 1989) have pollen characteristics that provide clues to the existence of pre-Cretaceous proangiosperms. Equisetosporites chinleanus and Cornetipollis reticulata are inaperturate, banded, tectum discontinuous in furrows, columellate; bands are psilate and nexine 3-layered with an intermediate lamellate transitional layer in Equisetosporites whereas bands are scalariformly reticulate, and nexine is uni-/bi-layered, and non-lamellate in Cornetipollis. Mono-, Di, Tri-, and Zona- Crinopollis: Proximally reticulate, distinctly columellate, distally foveoreticulate a foveolateŕpitted, exhibiting diminution of columellae and footlayer. "Apertures" distal, (sub-) equatorial or distal or/and lateral. Monocrinopollis: monosulc(ul)ate (2 compound sulc(ul)i), pontoperculum narrow; Dicrinopollis: bisulc(ul)ate with wide detachable operculum; Tricrinopollis: trisulc(ul)ate, sulc(ul)i independent, one distalsulcu(lu)s flanked by a pair of equatorial or lateralsulc(ul)i, ±parallel to the distal sulcus; Zonacrinopollis anasulcatus: zonasulcu(lu)s dividing the grain into 2 subequatorial halves. Pentecrinopollis traversei: sculpture clavate, with 5 distal aperturoid thinnings, finely reticulate proximally between the clavae. Polycolpopollis magnificus: zonasulculate to spiraperturate, coarsely reticulate.
Columellae supported coarse/fine reticulum and scalariform reticulum in the Crinopolles group and Cornetipollis respectively are assertive of their angiospermid nature; uniformly thickened endexine in some taxa of the Crinopolles may be indicative of the less evolved nature of sulc(ul)ate apertures and also of a desiccation minimizing adaptation during the arid Triassic Period.
The monosulculate to multiaperturoid character states of these palynomorphs had preceded the monosulcate > tricolpate > tricolporate > triporate evolutionary sequence of the Cretaceous. From the presence of columellae supported proximal reticulum, psilate tectum, proximo-distal luminal-foveoloid gradations (monocotyledonoid) and of endexine (dicotyledonoid) it is inferable that the Crinopolles types straddle both classes. The 3-layered nexine in Equisetosporites may be representative of an evolutionary stage of pollen-wall of proangIosperms.
Key-words- Angiospermid palynomorphs, Late Triassic, Biodiversification.
INTRODUCTION
Dilcher (1984) Temarked "Studies of fossils, their affinities, analysis of their characters in the context of related modem taxa including the establishment of homologies will continue to be an important aspect of angiosperm palaeobotany". Pollen characters are subject to parallelism, convergence and possible reversal, and the study of fine structural details of fossil pollen opens up possibilities of new sources of phylogenetic evidence (Davis & Heywood 1963).
The Late Triassic angiospermid foveoreticulate to scalariformly reticulate pollen (Cornet, 1979a) differing from the psilately banded "Equisetosporites" has been named as Cornetipollis reticulata by Pocock and Vasanthy (1988), the specific epithet underscoring the angiospermous character. Cornet (1989) proposed 7 new genera for the Late Triassic angiosperm-like pollen from the Richmond rift basin of Virginia.
We are here dealing with the distinctively columellate Triassic palynomorphs: Equisetosporites chinleanus Daugh., Cornetipollis reticulata Pocock
100 GEOPHYTOLOGY
& Vasanthy and the Crinopolles group (Cornet 1989). These pollen types may be representative of different levels of pollen characters (apertural and exinal evolution in angiosperms). But our attempts to find modem analogues for the Triassic angiospermid palynomorphs have resulted in matching certain characters of the Triassic pollen partly with those of the angiosperms (Table 2).
Contributions by Vasanthy and Pocock (1986), Pocock and Vasanthy (1988), Pocock et al. (1988), Cornet (1989), Vasanthy et at. (1990, 1991) and Cornet and Habib (1992) have addressed the difficult and long-standing question "How old are the angiosperms?". And in this paper on the Triassic angiospermid pollen we are re-evaluating this enduring question.
"Angiosperm Origins: Pre-Cretaceous vs Lower Cretaceous" is a much-debated subject of palaeobotanical interest. Defenders of Palaeozoic origin (Axelrod 1961) advocated that precursors to proto-angiosperms of Jurassic through Cretaceous were in existence by Perma-Triassic; those of Mesozoic origin (Axelrod 1970, Takhtajan 1969), postulated that angiosperms diverged from an ancestral alliance during the middle Mesozoic and those of Lower Cretaceous (Doyle 1969, Muller 1970) hypothesized on the bases of the then available fossil evidence that angiosperms were not present anywhere in the world before Barremian time and angiosperm fossil mostly leaves first become a regular component of fossil record during the late lower Cretaceous, approximately 100 million years ago.
Cornet's report (1979b) on the Late Triassic monosulcate, tectally perforate, columellate angiospermid palynomorphs received immediately a favourable citation: "even under the light microscope, many of the grains described by Cornet show exine features that occur today only in angiosperms; coarsely reticulate sculpture resulting from large perforations in the tectum (are) supported by well-developed rods or columellae rather than granules or partitions of alveolae" (Doyle 1978). Doyle and Hotton (1991) remarked that the Late Triassic reticulate-columellate pollen type described by Cornet (1989) as the Crinopolles group is reminiscent of Liliacidites and approved of a relationship between the Crinopolles and angiosperms.
The objectives of this paper are to emphasize the angiospermid features observed in the selected angiospermid Triassic pollen types (occurrence of columellae supporting either the scalariformly reticulate or coarsely to fmely reticulate or psilate tectum and the presence of endexine but unmodified under the "apertures" and to underscore the relationship between the angiosperm pollen and, the selected Triassic angiospermid palynomorphs. Their angiospermid pollen characters may be representing the evolutionary stages in angiosperm pollen wall constitution.
MATERIAL AND METHOD
Palynomorphs included in this work were recovered from the late Triassic sediments of USA Outgroup locality VB4 of the Richmond Basin, Virginia (Cornet 1989); earliest Carnian to late middle Carnian or early late Triassic; Petrified Forest National Park, Chinle, Arizona, and Ciniza lakebeds, Chinle Formation, Western New Mexico.
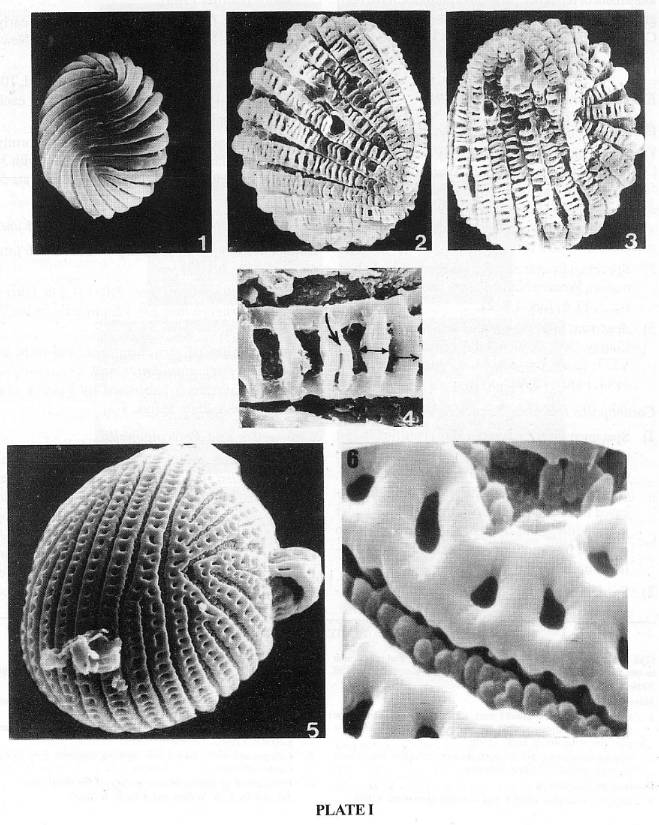
PLATE 1 (SEM)
Equisetosporites chinleanus (from the Ciniza lakebeds, Chinle Formation, Western New Mexico, Late Carnian).
1. Obliquely oriented psilate bands; note the two points of convergence of bands in this view. x 1000.
Cornetipollis cf. reticulata (from Middle Carnian of the Richmond basin, Virginia, VB4, VA, USA).
2-3. Two sides of a bilaterally and rotationaliy <90°) symmetric grain (resembling medially sectioned half). x l025.
4. Enlargement of 2; the ladder-like (scalariform) mural pattern, a bifurcating rung (arrow) and the distill doming or medial convexity of rung (double-headed arrow). x 4l00.
Sanchezia decora Leonard and Smith (Tribe Trichanthereae, Acanthaceae, from NY: New York Botanic garden Herbarium).
5. A rotationally symmetric grain; note the bifurcation of one of the three middle bands and the supra-oral flaps on the right side of the grain and the unusual presence of baculate elements (throw-back of a primitive character?) in the pseudocolpi. x l000.
6. Enlargement of 5; note the differences between the exine surface details of bands of Cornetipollis and Sanchezia. x 7000.
1-4 by J. W. Walker and 5-6 by S.A.J. Pocock.
EVOLUTION OF PROANGIOSPERMS DURING LATE TRIASSIC 101

102 GEOPHYTOLOGY
Palynomorph extraction methods and details of microscopic (LM, SEM and TEM) techniques and of lodgement of reference palynomorph slides, have been given in our earlier works (Pocock & Vasanthy 1988, Cornet 1989, Cornet & Habib 1992).
DESCRIPTION
Equisetosporites and Cornetipollis
Equisetosporites chinleanus Daugherty 1941
1) Specimen from the type locality (petrified forest member, Chinle
Formation, Arizona) late Carnian (Pocock & Vasanthy, 1988, Plates I, II, III).
Ellipsoidal) 48(50.5)56 x 25(31)39 um; ellipsoidal; psilate, 9(15)18 ectexinal bands
enveloping the smooth nexinal core.
2) Specimen from the Ciniza lakebeds, Chinle Formation, Western New Mexico, late Carnian
(pl. 1, fig.l, Pl.5, figs.1 & 2).
3) Specimen from the Chinle Formation) Garyfield County, Utah, Western USA (Zavada 1992,
Plate VI, 1, 2, dimensions 41 x 29 um and Zavada 1984, Figures 8, 9).
Cornetipollis reticulata Pocock & Vasanthy, 1988
1) Specimen from the type locality (petrified Forest member) Chinle
formation, Arizona), late Carnian; ellipsoidal, 63 (77±8) 86 x 31 (45±7) 60 um,
comprising an almost smooth central body encased in a banded exterior, bands (14±1),
wider in the mid-region, and tapering towards the two points of fusion, at times
manifesting slight twisting (pocock & Vasanthy 1988).
2) Specimens from Ciniza lake beds, Chinle Formation, Western New Mexico, late Carnian,
ellipsoidal, 90 x 75 um, 16-18 scalariformly reticulate bands (Cornet 1985).
3) Specimens from the palynoflorule VB4, early Carnian, the Richmond rift basin of
Virginia, Newark Supergroup:
a. Spheroidal, D= 108.0 um, 90° rotated, 10- 11 scalariformly reticulate bands in each hemisphere (Cornet & Habib 1992).
b. Ellipsoidal, 90 x 75 um, 10-11 scalariformly reticulate bands in each hemisphere with 3-4 middle bands (Plate 1, figs. 2-4, Plate 5, fig. 3).
Dimensions of lumina: Cornetipollis: 1.0-1.5 um. Columellar heights: Equisetosporites: 0.5-0.6 um; Cornetipollis: 0.5-1.0 um.
Nexine: Equisetosporites: 3.0-4.0 um (three layered); Cornetipollis: 3.0-3.5 um uni- or bi-(?) layered.
Description of structure and sculpture of Equisetosporites chinleanus and Cornetipollis reticulata have been published by Pocock and Vasanthy (1988, Plate 5, figs. 1-3).
Crinopolles and Polycolpopollis
Specimens and holotypes (3 species of Monocrinopllis; Dicrinopollis operculatus, 2 species of Pentecrinopollis, Zonacrinopollis anasulcatus and Polycolpopollis magnificus) are from the outcrop locality VB4 of the Richmond rift basin, Virginia, USA (Cornet 1989). Monocrinopollis microreticulatus has been reported (Litwin, 1985) from the upper Carnian Petrified Member of the Chinle Formation
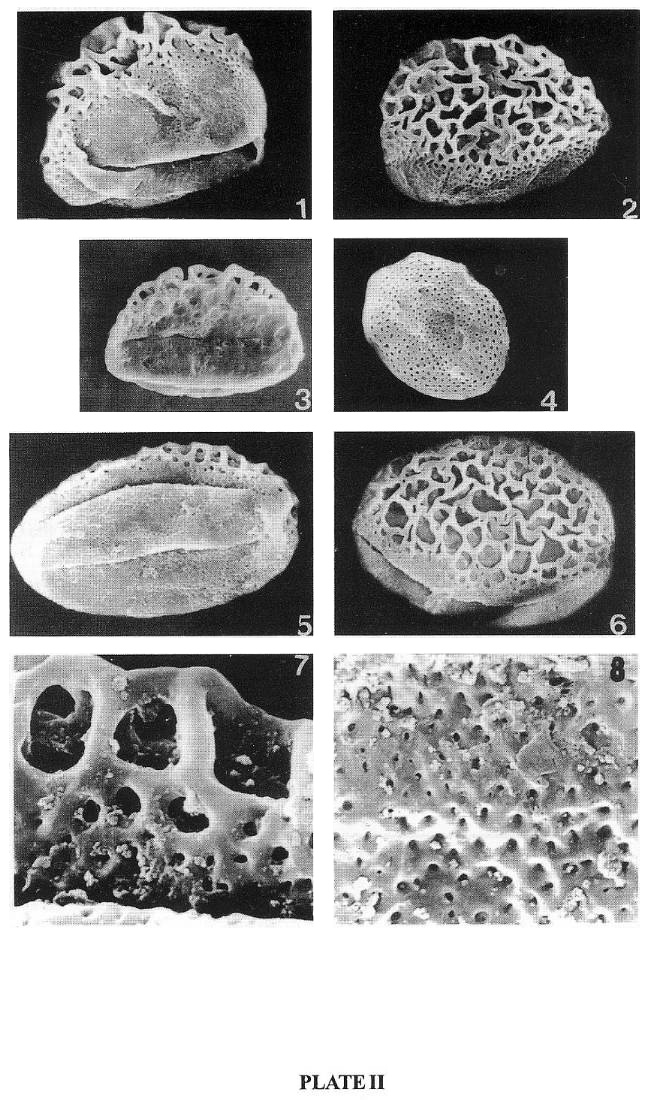
PLATE 2 (SEM)
(The taxa belonging to the Crinopolles group are from earliest Carnian to late middle Carnian or early late Triassic), the Richmond basin of Virginia, USA)
Monocrinopollis doylei
1. Oblique lateral view (44 x 35 um), showing transition from fine to coarse
sculpture; one of the two sulc(ul)i and the pontoperculum are in view. x 1000.
2. Oblique lateral view (48 x 35 um), showing transition from coarse to finely
graded reticulate sculpture.
Dicrinopollis operculatus
3. Oblique distal view (38 x 25 um) showing operculum. x 1000.
Monocrinopollis microreticulatus
4. Micro-reticulately patterned proximal (3l x 25 um) side. x 1000.
Tricrinopollis olsenii
5. Oblique distal view (62 x 38 um), showing median sulc(ul)us and one of the
lateral sulc(ul)i, x 1000.
6. Oblique proximal view (40 x 30 um) showing one of the lateral sulc(ul)i. x
l000.
7. Enlargement of the lateral side showing transition from fine to coarse reticulum.
8. Enlargement of foveoreticulate surface of the distal side.
1-2, 4-8 by J. W. Walker and 3 by R. R. Keith.
EVOLUTION OF PROANGIOSPERMS DURING LATE TRIASSIC 103
104 GEOPHYTOLOGY
(Petrified Forest national Park, Arizona).
Age: Pentecrinopollis traversei, P. gemmatus, Tricrinopollis minutus, Dicrinopollis operculatus, Zonacrinopollis anasulcatus and Polycolpopollis magnificus were in early Carnian; Tricrinopollis olsenii, Monocrinopollis doylei and M. walkeri were recovered from early to mid-Carnian and M. microreticulatus from early to late Carnian sediments. Presumptive Habitats: Pentecrinopollis spp., Tricrinopollis spp. and Monocrinopollis doylei were deposited in environments adjacent to rivers and tributaries, but not in basin swamp or lake environments. Tricrinopollis olsenii and Monocrinopollis doylei ventured into the coal swamp environment. Monocrinopollis mulleri, in addition to fluvial and deltaic environments, were also in silty "shoreface" environments, probably above and below bar-finger and distributory mouth bar sandstones - unstable shoreline areas around the large lakes that filled the rift basin. Monocrinopollis walkeri and M. microreticulatus have been recovered from depositional sequences different from other Crinopolles. M. walkeri occurs in delta environments, mainly those characterized by black shales interbedded with thin sandstones; M. microeticulatus occurs mainly in overbank shales rich in megafossil plants. Polycolpopollis magnificus probably grew in the wet coal forming environment of the Richmond basin.
Form of Corpus: Oblong to elliptical in Pentecrinopollis (rarely rounded or circular), Tricrinopollis (rarely sub-rounded), Dicrinopollis, and Zonacrinopollis; oblong to spherical, usually elliptical in Monocrinopollis and spherical to subspherical in Polycolpopollis Aperture Types,
Number and Configurations: Monocrinopollis - compoundly monosulc(ul)ate, i.e. two closely spaced sulc(ul)i separated by a narrow "pontoperculum"; occasional anomalous sulc(ul)i in equatorial position; Dicrinopollis - two sulc(ul)i located near lateral or equatorial sides of grain separated by a wide operculum; Tricrinopollis - normally with one distal sulcus flanked by a pair of lateral or equatorial sulc(ul)i; lateral sulc(ul)i oriented nearly parallel to the distal sulcus; occasionally one equatorial sulc(ul)us missing or an additional sulc(ul)us placed on the distal side (total four); Pentecrinopollis - pentasulc(ul)ate or penta plicate with five sulc(ul)i or aperture-like furrows (range 4-6 sulc(ul)i or furrows); Zonacrinopollis - zonasulc(ul)ate with an additional sulcu(lu)s on distal side oriented parallel to grain's longitudinal axis; Polycolpopollis - tricolp(oid)ate to zonasulc(ul)ate, usually spiraperturate.
[We leave to the reader the choice between sulcus (furrow centered at the distal pole) and sulculus (furrow more or less parallel to the equator either at the equator or displaced toward a pole, generally distal to the equator but not centered at the distal pole) to designate the aperture types of the Crinopolles group (Halbritter & Hesse 1993)].
Sculptural and tectal surface patterns: Crinopolles are
typically, but not exclusively, reticulate-collumellate:
e.g. large sexinal gemmae or prominent clavae whose heads are joined by a micro-reticulum
(lumina: <1.0 um) in P. traversei (pl. 3, fig. 4). Proximal
sculpture in Dicrinopollis, Tricrinopollis, and Zonacrinopollis is coarsely
reticulate and that in Monocrinopollis. is finely to coarsely reticulate. Their
distal exines are foveo-reticulate (Zonacrinopollis) to foveolate
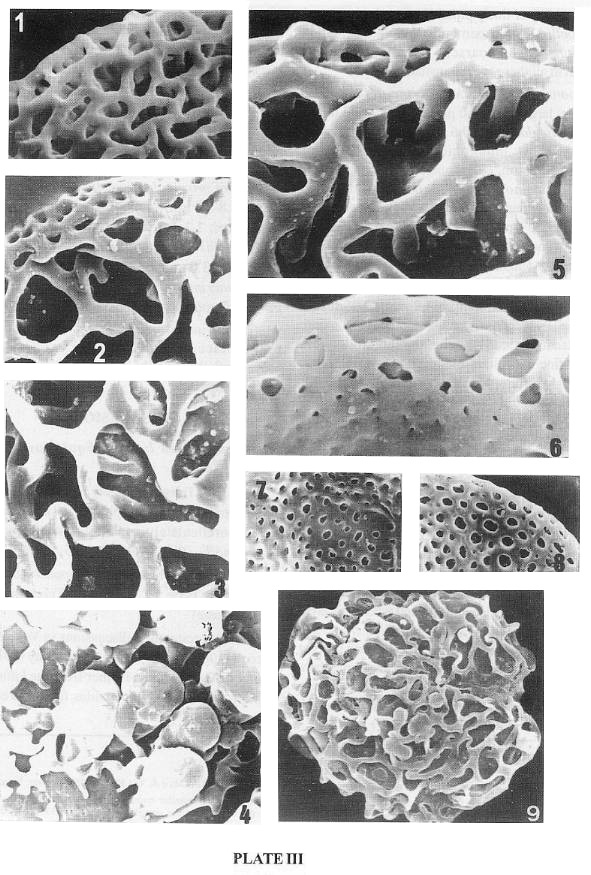
PLATE 3 (SEM)
Monocrinopollis mulleri
1-2. Enlargements of proximal reticulum supported by columellae.
Monocrinopollis doylei
3. Enlargement of proximal reticulum supported by columellae.
Pentecrinopollis traversei
4. Enlargement of proximal side; note the imperfect reticulum joining the clavae at
about the junction of neck and head, which may be uniquely "angiospermous".
Tricrinopollis olsenii
5. Enlargement showing proximal reticulum supported by relatively tall columallae.
6. A part of the distal side enlarged that shows the reduction of lumina and loss of
columellae as the footlayer rises to meet the tectum.
M. microreticulatus
7-8. Enlargement showing micro-reticulum of the proximal side.
Polycolpopollis magnificus (79 x 73 um).
9. Portions of sulcu(lu)s visible; large reticulum supported by columellae.
1-9 by J. W. Walker.
EVOLUTION OF PROANGIOSPERMS DURING LATE TRIASSIC 105
106 GEOPHYTOLOGY
(Tricrinopollis) or finely pitted (Monocrinopollis and Dicrinopollis). The proximo-distal surface pattern in Polycolpopollis is coarsely reticulate. Muri of reticulum in all these taxa are psilate.
Dimensions of lumina: Monocrinopollis doylei: dimorphic, larger: 4.5-12.0 um; smaller: 0.8-2.0 um; M microreticulatus: 0.4-0.6 um; M. mulleri: 1.5-6.0 um; M. walkeri: dimorphic, larger: 1.1-2.6 um; smaller: 0.4-0.5 um, Dicrinopollis operculatus: 3.0-9.8 um; Tricrinopollis olsenii: heteromorphic reticulum, larger lumina 3.0-10.5 um; Pentecrinopollis traversei: <1.0 um; Zonacrinopollis anasulcatus: 4.5-7.5 um and Polycolpopollis magnificus: 3.0-11.0 um (maximum length of lumina).
Exine structure: Monocrinopollis spp.: 2-layered, with a well-developed footlayer proximally, ectexine thinner distally with footlayer discontinuous or missing, columellae reduced to granules and short rods under area with finer reticulum, endexine if present is thicker under distal aperture. Monocrinopollis doylei: 1.8-3.0 um; M. microreticulatus: 0.4-0.6 um; M mulleri: 1.1-1.5 um; M. walkeri: 0.6-0. 7 um; Dicrinopollis operculatus: ca. 1.4 um with a well-developed footlayer proximally, ectexine thinner distally, with a footlayer discontinuous or missing and columellae reduced or absent under area with finer reticulum, endexine present; Tricrinopollis olsenii: 2.0-3.0 um, with a well-developed footlayer proximally, distal ectexine much thinner than proximal ectexine, footlayer 0.15-0.21 um thick proximally, disappears equatorially and distally under finer reticulum, endexine present, continuous and noticeably thicker under sul(cu)li and where footlayer is absent (Plate 5: 6); Pentecrinopollis traversei: exine composed of a 1.0-1.5 um thick nexine bearing large prominent clavae whose heads are joined by a delicate reticulum that abruptly becomes an imperforate tectum along the sides of furrows containing the sulculi (Cornet 1989); Zonacrinopollis anasulcatus: exine 2-layered with a well-developed foot layer proximally, ectexine thinner distally with footlayer discontinuous or missing and columellae reduced or absent under area with fine reticulum, endexine not observed; ca. 3.0 um and Polycoipopollis magnificus: 4.0-6.0 um, nexine ca. 0.8-1.0 um thick, apparently single-layered without an endexine, sexine thins at apertural margins as reticulum joins footlayer.
Heights of proximal columellae: Monocrinopollis doylei: 1.8-3.0 um; M. microreticulatus: 0.4-0.6 um; M. mulleri: 1.1-1.5 um; M. walkeri: 0.6-0.7 um; Dicrinopollis operculatus: ca. 1.4 um; Tricrinopollis olsenii: 2.0-3.0 um; Pentecrinopollis traversei: <0.5 um; Zonacrinopollis anasulcatus: ca. 2.2 um and Polycoipopollis magnificus: 4.0-6.0 um.
Table 1 includes the dimensions of the selected pre-Cretaceous columellate angiospermid palynomorphs and their key characters for easy comprehension.
DISCUSSION
Inaperturate and banded Equisetosporites vs Cornetipollis
The Late Triassic psilately banded Equisetosporites is exo- and endo- morphologically different from the foveoreticulately and/or scalariformly banded Cornetipollis (Pl. 1, figs 1-4, Pl. 5, figs 1-3).
"Reticulately" banded Cornetipollis reticulata vs the pollen type of the tribe Trichanthereae. (Acanthaceae).
Without reiterating discussions in Vasanthy and Pocock (1986) and Pocock and Vasanthy (1988), we briefly differentiate the former from the latter (Pl. 1, figs 2-6, PI.5, figs 3-4): besides the differences in
PLATE 4 (SEM)
Classopollis minor Pocock & Jansonius 1961
1. Distal view, paral membrane detached exposing the edges of endo-annulus; note the
sub-equatorial furrow (white arrow). x 3600. (SEM by S.A.J. Pocock).
Classopollis harrisii Muir & Van Kon. Middle-Jurassic:
Rhaeto-Liassic.
2. Note the columellar complexity (C), faint lamellations within the nexine and the
complex tectum (T). x l3100 (by J.Medus).
EVOLUTION OF PROANGIOSPERMS DURING LATE TRIASSIC 107
108 GEOPHYTOLOGY
Table 1. Key characters of the selected pre-Cretaceous columellate angiospermid palynomorphs
| Taxa | Key characters | ||
| Daugherty (1941) | Dimension (lxb) | No. & type of apertures | Surface features |
| 1. Equisetosporites chinleanus Pocock & Vasanthy (1988) |
48-56 x 25-39 um | 9-18 furrows | psilate |
| 1. Cornetipollis reticulata Cornet (1989) |
63-86 x 31-60 um | 14-15 furrows | scalariformly reticulate |
| 1. Monocrinopollis | 1-distal compound sulculus, pontoperculum narrow |
proximo-distally finely or coarsely reticulate to foveoreticulate or faintly pitted |
|
| a. M. doylei | 36-51 x 29-45 um | ||
| b. M. microreticulatus | 27-35 x 21-27 um | ||
| c. M. mulleri | 36-49 x 25-40 um | ||
| d. M. walkeri | 24-38 x 16-35 um | ||
| 2. Dicrinopollis operculatus | 38-46 x 25-39 um | 2-lateral sulculi with a wide foveoloidate operculum |
proximo-distally coarsely reticulate to foveoreticulate |
| 3. Tricrinopollis olsenii | 42-53 x 25-39 um | 1-distal + 2 lateral sulculi | proximo-distally coarsely reticulate to foveoreticulate |
| 4. Pentecrinopollis traversei | 66-88 x 42-55 um | "5-plicae" or 5-sulculi | clavae on micro-reticulum |
| 5. Zonacrinopollis anasulcatus | 39-41 x 30-33 um | zonasulculus | proximo-distally coarsely reticulate to foveoreticulate |
| 6. Polycolpopollis magnificus | D: 69-90 um | zonasulculus, spiraperturate | coarsely reticulate |
dimensions, number of bands, tectal columellar distribution patterns, and the presence of baculoid sculptural elements (Pl. 1, figs 2-5), we underscore here the following exinal differences of significant diagnostic value: the columellae of C. reticulata are short (0.5 um) and well-fused with the foot layer or unstructured nexine whereas the basally branched or "hollowed out" columellae (3.5-4.0 um) of Trichanthereae type of pollen are superficially "attached" to the surface of endexine or nexine which
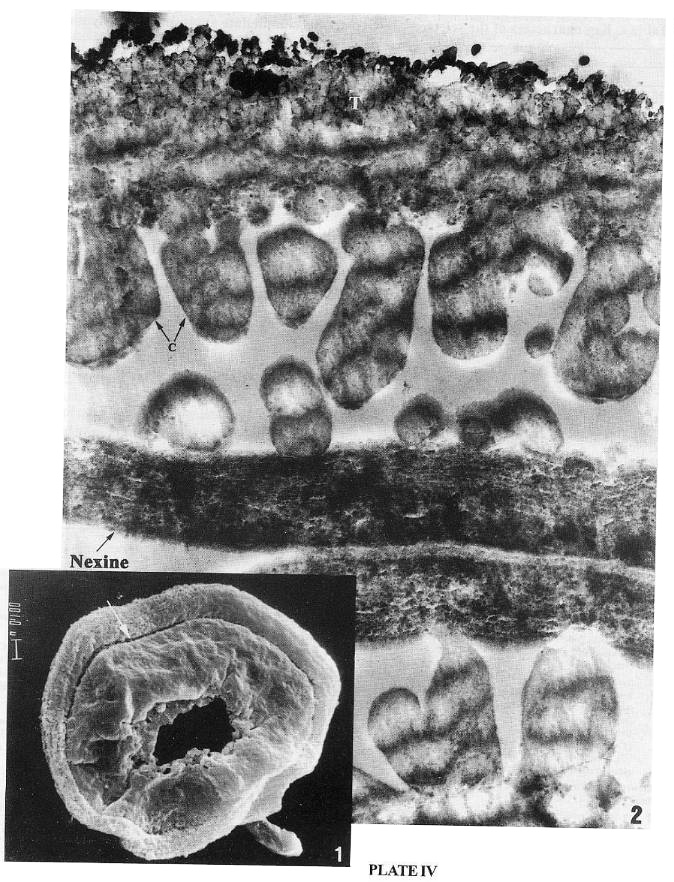
PLATE 5 (SEM)
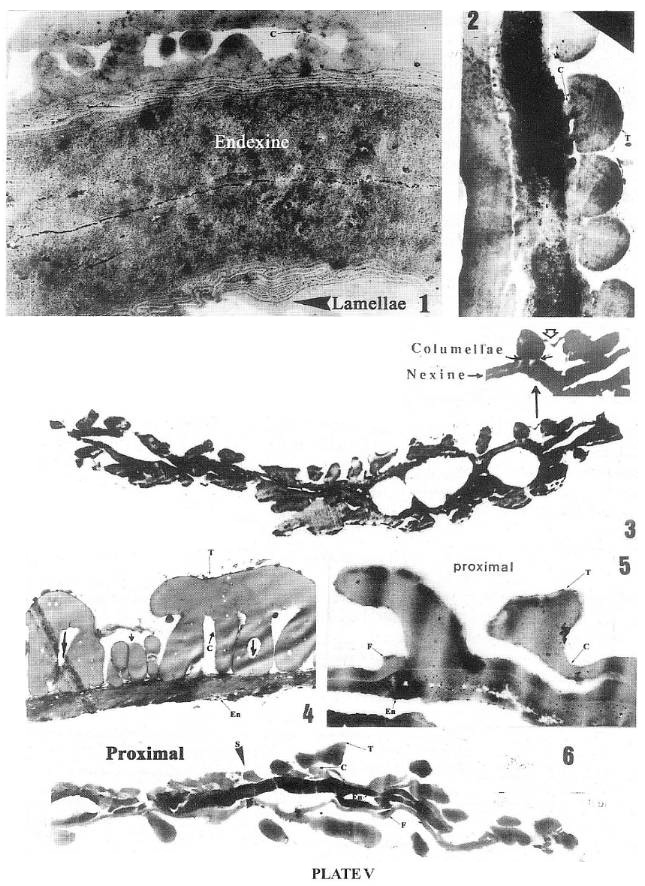
Equisetosporites chinleanus, palynomorph from the Ciniza lakebeds, Chinle Formation, Western Mexico, late Carnian.
1. TEM section of Pl. 1, fig. 1 illustrating the juxtaposed layers of endexine bordered by their respective lamellate zones; note the periclinal lamellae darkly staining and interlamellae (weakly staining) are undulate, convoluted, looped and also humped (arrow). x 21000.
2. Section through about half of a grain with rotated halves or hemisphere (<90° rotation of bands): a parallelly sliced band in the left side and five cross sectioned bands in the right: the inner faces of the opposing hemispheres are juxtaposed due to the flattening of the grain or its lateral compression. The thick tectum (T) hemispherical in cross section is supported by short columellae (C), mostly of inclined or oblique orientation arising from the non-lamellated footlayer. x 4900.
Cornetipollis cf. reticulata, from the Middle Carnian of the Richmond. basin (VB4), VA, USA.
3. A thin bilaterally compressed section of the palynomorph in Plate 1: 2 & 3 passing through the rotated furrows and bands (across lumina or foveolae, and marginal cross-walls or muri) exhibiting different planes of sectioning. x 4250. Inset: Enlargement (x 6000) of the upper right portion of the above section; note the columellae (arrows) and conic and wedge-shaped sections of cross-walls interlinked by a thin tectal connection of part of marginal mums (short white arrow).
Sanchezia equatoriensis Leonard (Tribe Trichanthereae, Acanthaceae) from the NY Botanic Gardens Herbarium.
4. The basally bifurcated or hollowed (arrow) columellae (C) supporting the thick tectum (T) are supeficially "attached" to the surface of endexine or nexine which encloses many small "gaps" and is lined by a few lamellae in the upper part. x 27500.
Monocrinopollis doylei
5. TEM enlargement of exine showing dark non-laminated endexine (En) with many small holes or "gaps" and a footlayer (F) on the proximal side; note the columellae (C) and the tectum (T) of coarsely reticulate proximal side. x 27470
Tricrinopollis olsenii
6. TEM showing coarsely reticulate-columellate proximal side (on bottom and upper right) and foveolate distal side (upper left); note the darker endexine, and the ectosulc(ul)us (S). x 4,690.
1-2 by A.V. Chadwick; 4 by J.R. Rowley. and 5-6 by J.W. Walker;
EVOLUTION OF PROANGIOSPERMS DURING LATE TRIASSIC 109
110 GEOPHYTOLOGY
encloses many small "gaps" and lined by a few lamellae in the upper part (Pl. 5, fig. 4).
We here underscore that foveoreticulate bands occur not only in extant Trichanthereae pollen and their fossil "look-a-likes" from the Late Triassic, Late Jurassic, Cretaceous and Tertiary but also in the Upper Cretaceous non-columellate spore Costatafoveosporites foveolatus Deak (Srivastava 1990).
Change of symmetry
Radial through rotated symmetry via torsion which is probably a non-adaptive character, has been encountered in plant groups of different geological ages and evolutionary.lineages: Tornopollenites complex and Ephedripites complex (Triassic through Tertiary), and recent taxa Ephedra spp. (Ephedraceae), Holochlamys (Araceae in Thanikaimoni 1969) and acanthaceous Bravaisia, Sanchezia, Trichanthera (Tribe Trichanthereae) and Nilgirianthus (Pocock & Vasanthy 1986 and references cited therein).
Columellae supported reticulate exine
Columellate infratectum and non-lamellate (non-gymnospermous), unilayered nexine in combination
Table 2. Similarities between the selected Triassic angiospermid palynomorphs and angiosperm pollen
| A. Triassic angiospermid palynomorphs (1) |
Characters of A "analogous" with those of B |
B. Partly comparable pollen types in angiosperms |
| Crinopolles Group | a) coarser reticulum grading into finer toward distal apertures; b) dimorphic coarser reticulum in some taxa; c) psilate muri of reticulum; d) thin footlayer of non-apertural region | Monocots (2) |
| Crinopolles Group | a) reticulate - columellate exine structure; b) two or more sulci(ul)i* or furrows on the distal and equatorial sides; c) dimorphic luminal pattern | Liliaceae (2) |
| Monocrinopollis | compound distal sulc(ul)us* with pontoperculum | Monocots (3, 4, and 5) |
| Dicrinopollis operculatus | a pair of widely spaced sulc(ul)i* with a detachable operculum | a number of different monocot genera, e.g. Xyris - Xyridaceae (3); Polianthes - Agavaceae (6) |
| Tricrinopollis | tricolpoidate or trisulc(ul)ate* | Nelumbo - Nymphoideae (7) |
| Zonacrinopollis | occasional union of eq sulc(ul)i* | Nelumbo - Nymphoideae (7) |
| Zonacrinopollis | zonasulc(ul)ate* | Monocots (3) and primitive dicots (8) |
| P. traversei | large size and finely reticulate exine | insect pollinated angiosperms (1) |
| Polycolpopollis magnificus | ring sulc(ul)ate* or spiroaperturate | Scrophulariaceae, Berberidaceae, and Acanthaceae (9) |
* Sulcoidate apertures of the Triassic Crinopolles group are partly similar to the sulcate apertures of angiosperms.
(1) Cornet, 1989; (2) Walker and Walker, 1984; (3) Thanikaimoni, 1969; (4) Chanda and Ghosh, 1976; (5) Chanda et al., 1978; (6) Alvarez and Köhler, 1989; (7) Kuprianova, 1979; (8) Walker, 1976; and (9) Furness, 1985.
EVOLUTION OF PROANGIOSPERMS DURING LATE TRIASSIC 111
with a foveo-reticulate to scalarifomlly reticulate tectum in Cornetipollis reticulata strongly impart an angiospermid semblance, despite the lack of distinctive germinal aperture in the nexine. It is generalised that the earliest unequivocal angiosperm pollen grains from the Lower Cretaceous (Barremian-Aptian) have reticulate sculpturing with tectate-columellate wall structure (Zavada 1984). Zavada hypothesized that in angiosperm pollen the reticulate sculpturing is indicative of sporophytic self-incompatibility (SSI) while imperforate and microperforate exine sculpturing is indicative of gametophytic self-incompatibility (GSI). Applying the same criteria, could we infer that the Late Triassic Cornetipollis and Crinopolles were sporophytically self-incompatible?
Pollinators of pre-Cretaceous Period
The pollination process seems to be the principal driving force in angiosperm evolution that might have led to many and marvellous convergences between plant and pollinators (Gottlieb 1968). According to Takhtajan (1969) the first pollinators of the angiosperms must have existed long before the Cretaceous; honeybees known from Tertiary deposits probably arose in the Cretaceous period and therefore beetles remain as the most likely pollinators of extinct angiosperms.
Endexine thickening in Mono-, Di-, and Tri-Crinopolles structure:
Proximal exine is columellate with a thicker footlayer in Monocrinopollis, Dicrinopollis, Tricrinopollis, and Zonacrinopollis; distally columellae are much reduced to granules or short rods and footlayer is absent. Endexine is thickened under aperture(s) (e.g. Tricrinopollis olsenii in Pl. 5, fig. 5) in the three aforementioned taxa. Diminution of columellae and footlayer (ectexinal) and thickening of endexine at the furrows may be functionally semi-harmogathic or desiccation minimising adaptation protecting the pollen cytoplasm (Heslop-Harrison 1979) under the arid climatic condition of the Triassic.
Evolution of columellae
If the columellae are the exclusive features of angiosperms, then how to account for columellar and tectal complexity in Mesozoic coniferalean Circumpolles? (Plate 4, fig. 2). Pre-Cretaceous Classopollis has a columellate infrastructure but has an unusual apertural arrangement and the exinal elaborations (Pocock et al. 1990).
The angiosperm-like pre-Cretaceous pollen Cornetipollis is tectate-columellate with a thin foot layer and lacks a sulcus but with multiaperturoidate grooves. The hypothetical view on the primitivity of angiospermic exine columellae is not only supported by their presence in the Cretaceous angiosperm pollen (Doyle & Hickey 1976) but also in these reticulately sculptured pre-Cretaceous angiospermid palynomorphs.
Phytogeographical origin of earlier diversified Monocotyledons
Based on the preponderance of both neo- and palaeo- phytogeographical evidences, Walker & Walker (1986) presumed that cladistically ancient (Doyle 1973) Monocotyledons had a laurasian (north temperate) origin rather than Gondwanian (continent connecting India with S. America, S. Africa, Antarctica and Australia from Carboniferous to Jurassic). But the prevalent assumption is that the monocots like the dicots had a basically Gonwana origin. It is conjectural that pre-Cretaceous pollen exhibiting trends towards monocot diversification had a laurasian origin as evidenced from the presence of a few monocotyledonid pollen character states in the selected Crinopolles taxa from Richmond Basin, Virginia, USA (Table 2).
The tectate-columellate wall structure is observed in most primitive families and orders of monocots. Endexine is very rare in monocots and when present is different from the endexine observed in dicots (Zavada 1983). That the endexine in the Crinopolles (Pl. 5, figs 5,6) is dicotyledonid rather than monocotyledonid is conjectural. So the pollen types of the Crinopolles group seem to straddle both the mono- and di- cotyledonous classes.
112 GEOPHYTOLOGY
Aperture evolution
The hypothetically most primitive monosulcate pollen characterises Magnoliales, some other primitive dicots, the majority of monocots and many gymnosperms including Bennettitales, cycads, Welwitschia, and Ginkgo. The monosulcoid to multiaperturoid (Table 1) character states of these palynomorphs appear to have preceded the monosulcate-> tricolpate-> tricolporate aperture evolution of the Cretaceous.
Pseudoapertures or harmomegathic furrows in Cornetipollis
and Equisetosporites and sulcoidate apertures ofCrinopolles lacking localised
germinal pore with or without endexinal thickening had probably provided exits by flexure
for pollen tube.
Triassic Crinopolles, upper Albian Liliacidites and extant Lilium pollen:
Dissimilarities
Comparative exine structural analysis of Late Tricrinopollis and Monocrinopollis (pl. 5, figs. 5 & 6) differ from Liliacidites (TEM in Walker & Walker, 1984) and Lilium longiflorum (Baldi et al., 1987) not only by their tall columellae and considerably thicker tectum but also by the presence of a thick endexine or nexine-2. Presence of end exine is not typical of monocotyledons
Inferences
1. Striations-like-apertures underlined by unmodified endexine and/or
nexine are indicative of pollen-wall protective strategy (desiccation minimizing) for
pollen protoplasm during anther-stigma transits in the arid climate of the Triassic.
2. Three layered nexine in Equisetosporites may be representative of an
evolutionary stage in nexine evolution (Guedes 1982; Vasanthy et al. 1990).
3. Proximo-distally reticulate to foveo-reticulate exine in Crinopolles group is
apparently monocotyledonoid.
4. Reticulate-columellateeXines in Cornetipollis and Crinopolles
group are assertive of their angiospermic nature.
5. The Crinopolles types seem to straddle both monocots and dicots.
6. The Late Triassic angiospem1 precursor (relatively "fragile") plants which produced Equisetosporites, Cornetipollis and Crinopolles might have been probably non-fossilized due to the aridity-induced degradation of organic matters of vegetative and reproductive parts except exines formed of one of the hardest organic compound (the carotenoid "sporopolenin").
ACKNOWLEDGEMENT
We are grateful to J .R. Rowley, A. V. Chadwick, J. W. Walker, R.R. Keith and J. Medus for providing SEM and TEM photographs.
REFERENCES
Alvarez, A & Kohler, E 1989. Morfologia del polen de las Agavaceae y algunos generos afines. Grana 26:
25-46.
Axelrod, DI 1961. How old are the angiosperms? Amr. J. Sci. 259: 447-457.
Axelrod, DI 1970. Mesozoic paleogeography and early angiosperm history. Bot. Rev. 36: 277-319.
Baldi, BG, Franceschi, YR, & Loewus, FA 1.987. Localisation of phosphorus and cation reserves in Lilium
longiflorum. Plant Physiol. 83: 1018-1021.
Chanda, S & Ghosh, K 1976. Pollen morphology and the evolutionary significance of the Xanthorrhoeceae. In:
Ferguson, I.K. & Muller, J. (eds). The evolutionary significance of the exine. Linn. Soc: Symp. Ser. 1:
527-539. Academic Press Inc. New York.
Chanda, S, Lugardon, B & Thanikaimoni, G 1978. On the ultrastructure of pollen aperture in Calectasia R. Br.
(Xanthorrhoeceae). Pollen Spores 20: 351-365.
Cornet, B 1979a. Noteworthy palynomorphs. ICP Inf. Newsl., 2(2): 3.
Cornet; B 1979b. Angiosperm-like pollen with tectate-columellate wall structure from the upper Triassic and
Jurassic of the Newark supergroup, USA (Abs.). Palynology 3: 281-282.
Cornet, B 1989. Late Triassic angiosperm-like pollen from the Richmond rift basin of Virginia, U.S.A.
Palaeontographica 213: 37-87.
Cornet, B & Habib, D 1992. Angiosperm-like pollen from the ammonite dated Oxfordian (Upper Triassic) of
France. Rev. Palaeobot. Palynol. 71: 269-294.
Daugherty, LH 1941. The Upper Triassic flora of Arizona. Carnegie Inst. Wash. Bull. 526: 1-108.
Davis, PH & Heywood, VH 1963. Principles of Angiosperm taxonomy. Krieger, New York, 588 pp.
Dilcher, D 1984. Historical perspective of angiosperm evolution. Ann Mo. Bot. Gard. 71: 348-350.
Doyle, JA 1973. Fossil evidence on early evolution of the monocotyledons: their evolution and comparative
biology. Quat. Rev. BioI. 48: 399-413.
EVOLUTION OF PROANGIOSPERMS DURING LATE TRIASSIC 113
Doyle, JA 1978. Origins of angiosperms. Annu. Rev. Ecol. Syst. 9: 365-392.
Doyle, JA & Hickey, LJ 1976. Pollen and leaves from the mid-Cretaceous Potamac group and their bearing on
early angiosperm evolution. In: Beck, C. B. (ed.). Origin and early evolution of angiosperms.
Columbia University Press, New York and London. pp. 139-206.
Doyle, JA & Hotton, CL 1991. Diversification of early angiosperm pollen in a cladistic context. In: Blackmore,
S. & Barnes, S.H. (eds). Pollen and Spores: Patterns of Diversification. Clarendon Press, Oxford.
pp. 169-195.
Furness, CA 1985. A review on spiraperturate pollen. Pollen Spores 27: 307-320.
Gottlieb, JE 1968. Plants: Adaptation through evolution. Affliated East-West Press Pvt. Ltd. 11.4 pp.
Guedes, M 1982. The stratification, ectexine structure and angiosperm evolution. Grana 21: 161-170.
Halbritter, H & Hesse, M 1993. Sulcus morphology in some monocot families. Grana 32: 87-99.
Heslop-Harrison, J 1979. Pollen walls - an adaptive system. Ann. Miss. Bot. Gard. 66: 813-829.
Kuprianova, LA 1979. On the possibility of the development of tricolpate pollen from monosulcate. Grana 18:
1-4.
Litwin, RJ 1985. Fertile organs and in situ spores offems from the Late Triassic Chinle formation of Arizona and
New Mexico with dicussion of the associated dispersed spores. Rev. Palaeo. Palynol. 44: 101-146.
Muller, J 1970. Palynological evidence on early differentiation of angiosperms. Bio Rev. Cambridge Phil. Soc.
45: 417-450.
Pocock, SAJ & Vasanthy, G 1988. Cornetipollis reticulata, a new pollen with angiospermid features from
Upper Triassic (Carnian) sediments of Arizona (U.S.A.), with notes on Equisetosporites. Rev.
Palaeobot. Palynol. 55: 337-356.
Pocock, SAJ, Vasanthy, G & Venkatachala, BS 1990. Pollen of Circumpolles - an enigma or morphotrends
showing evolutionary adaptation. Rev. Palaeobot. Palynol. 65: 179-193.
Srivastava, SK 1990. Dinocyst biostratigraphy of Cenomanian-Coniacean Formations of Western Gulf coastal
plain, southern United States. Palaeobot. 39: 155-235.
Takhtajan, A 1969. Flowering plants, Origin and dispersal. Oliver & Boyd: Edinburgh. 310 pp.
Thanikaimoni, G 1969. Esquisse palynologique des Aracees. Trav. Sect. Sci. Tech. Inst. Fr: Pondichery. 5:
29 p.
Vasanthy, G & Pocock, SAJ 1986. Radial through rotated symmetry of striate pollen of the Acanthaceae. Can.
J. Bot. 64: 3050-3058.
Vasanthy, G, Venkatachala, BS & Pocock, SAJ 1990. The evolution of angiospermid pollen characteristics:
Conjectures and Queries. In: Jain, K.P. & Tiwari, R.S. (eds). Proc. Symp. Vistas in Indian
Palaeobotany. Palaeobotanist 38: 131-146.
Vasanthy, G, Pocock, SAJ & Venkatachala, BS 1991. A comparative study of the Triassic Cornetipollis
reticulata and the tribe Trichanthereae of Acanthaceae. Poster Abstract of the 23rd AASP Annual
Meeting. Palynology 15: 254.
Walker, JW 1976. Comparative pollen morphology and phylogeny of the ranalean complex. In: Beck, C.B.
(ed.). Origin and early evolution of angiosperms. Columbia University Press, New York & London.
pp. 141-199.
Walker, JW & Walker, AG 1984. Ultrastructure of Lower Cretaceous angiosperm pollen and the origin and the
early evolution of flowering plants. Ann. Missouri Bot. Gard. 71: 464-521.
Walker, JW & Walker, AG 1986. Ultrastructure of Early Cretaceous angiosperm pollen and its evolutionary
implications. Pollen and Spores, form and function. S Blackmore & IK Ferguson (eds). Academy Press,
London. pp. 203-218.
Zavada, MS 1983. Comparative morphology and monocot pollen and evolutionary trends of apertures and wall
structure. Bot. Rev. 49: 311-379.
Zavada, MS 1984. The relationship between pollen exine sculpturing and self-incompatibility mechanisms. Pl.
Syst. Evol. 147: 67-78.
Zavada, MS 1990. The ultrastructure of three monosulcate pollen grains from the Triassic Chinle Formation,
Western United States. Palynology 14: 41-51.